In addition to being used as a dye, cobalt is also important to human nutrition as it is an essential part of vitamin B 12.Įstimated Crustal Abundance: 2. It has a relatively high melting and boiling point, the melting point being 1,495 degrees Celsius or 2,723 Fahrenheit and the boiling point being 2,927. Cobalt boride is produced under high temperature such as 1500 ☌. Some of these compounds are known as: cobalt blue, ceruleum, new blue, smalt, cobalt yellow and cobalt green. Cobalt-60 has a half-life of 5.27 years and decays into nickel-60 through beta decay.Ĭobalt compounds have been used for centuries to color porcelain, glass, pottery, tile and enamel. 
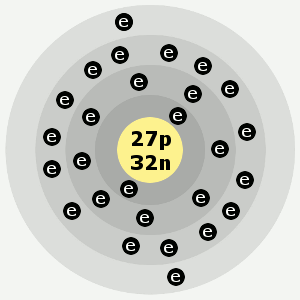
Cobalt is also used to make alloys for jet engines and gas turbines, magnetic steels and some types of stainless steels.Ĭobalt-60, a radioactive isotope of cobalt, is an important source of gamma rays and is used to treat some forms of cancer and as a medical tracer. Melting Point: 1330 ☌ (2430 ☏) Boiling Point: N/A: Density: 10 g/cm3: Solubility in H2O: N/A: Electrical Resistivity-6 10x -m: Poissons Ratio: 0.29: Specific Heat: 390 J/kg-K. Stellite alloys, which contain cobalt, chromium and tungsten, are used to make high-speed and high temperature cutting tools and dyes. Alnico, an alloy consisting of aluminum, nickel and cobalt is used to make powerful permanent magnets. Cobalt is usually recovered as a byproduct of mining and refining nickel, silver, lead, copper and iron.Īlthough cobalt is used in electroplating to give objects an attractive surface that resists oxidation, it is more widely used to form alloys. Cobalt's primary ores are cobaltite (CoAsS) and erythrite (Co 3(AsO 4) 2). Cobalt Liquid density at m.p., 7.75 g/cm Melting point, 1768 K (1495 C, 2723 F) Boiling point, 3200 K (2927 C, 5301 F) Heat of fusion, 16.06 kJ/mol. What is the melting point of cobalta)1449 Kb)1559 Kc)1669 Kd)1769 KCorrect answer is option D. Melting point refers to a particular point of temperature at which the element start. 
Cobalt boiling point The boiling point of Cobalt is 2927 ☌ (3200 K, 5301 ☏). Melting point refers to a particular point of temperature at which the element start changing its state from solid to liquid. Brandt was attempting to prove that the ability of certain minerals to color glass blue was due to an unknown element and not to bismuth, as was commonly believed at the time. Quick links for Chemical Engineering exam. The melting point of Cobalt in different unit is 1495 C (1768 K, 2723 F). The melting point of Cobalt in different unit is 1495 ☌ (1768 K, 2723 ☏). It also can be used in dental care.Cobalt was discovered by Georg Brandt, a Swedish chemist, in 1739. It is also used in medicine, in electroplating, and in agriculture.Ĭobalt(II) fluoride, a compound with the formula CoF2, is used as a catalyst to alloy metals. cobalt, Co nickel, Ni copper, Cu The table shows the melting point and density of some transition elements, compared to three metals in the periodic table that are not transition elements. Cobalt can be used in the production of catalysts too. Cobalt had been used in imparting a blue color to glazes, glass, and ceramics. Cobalt compounds are used in the manufacture of porcelain enamels and batteries as well as in paints and inks. Cobalt blue has been used as a coloring agent in jewelry and Chinese porcelain. Cobalt is ductile and ferromagnetic, and has a metal. It also alloys with aluminum, platinum, iron, and nickel. Cobalt, chemical symbol Co, atomic number 27, melting point 1495, is a high melting point metal. Uses of CobaltĬobalt, a chemical element with the symbol Co, is used in making magnets. Due to its high strengths, the alloys of this metal are used in aircraft production, etc. Cobalt nitrate is the inorganic compound with the formula Co (NO 3) 2. With industrial purposes, cobalt is usually used in alloys with iron, nickel, and aluminum, to produce powerful magnets. If consumed in larger doses, cobalt is very carcinogenic. This element is crucial for all living organisms, but in micro doses. This element can be found in some minerals, as it was discovered that there are some cobalt deposits at the bottom of the oceans. 1660, and viscosity measurements by the oscillating crucible method at. This metal used to be one of the materials widely used in ancient China to produce porcelain and add a bit of beautiful blue color to it. Physical Characteristics: melting point 254-256 (dec), boiling point 120/0.5mm (subl.) Related Categories: CVD & ALD Precursors Metal Beta-diketonates. maximum bubble pressure method at temperatures between the melting points and. Its name is believed to take roots from a German word meaning goblin. Cobalt is a light grey and bluish metal, which is similar to iron in its qualities and can be easily magnetized. CHEMICAL NAME : Cobalt (II) chloride CAS REGISTRY NUMBER : LAST UPDATED : 199710 DATA ITEMS CITED : 44 MOLECULAR FORMULA : Cl2-Co MOLECULAR WEIGHT : 129. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
